When It Comes To Improving Efficiency, Profitability And Sustainability In A Challenging Market, The Suppliers Of Mining Chemicals Have More Than A Few Tricks Up Their Sleeve.
By Jonathan Rowland
As is often noted in the pages of mining trade publications, the industry is facing a broad range of challenges. Miners need to extract more to meet growing demand for metals and minerals from a growing global economy, but do so from lower-quality, more complex ores containing more difficult contaminants, all while reducing costs and environmental impacts.
Amid the ink spilled, however, the role of mining chemicals is not often discussed. Yet developments in these unheralded elements of the mining process present significant opportunities to help solve some of the most pressing issues facing miners today. As Eammon Guitard, regional sales director at Solvay, told North American Mining: “The need for more high-performance reagents has never been greater.”
Finding Value in Lower Grades
Lower grades pose a number of difficulties for miners, from complex mineralogy, particularly when it comes to gangue materials, greater frequency of penalty elements, more complex water chemistry and higher pulp viscosities.
This is particularly true in areas where there are significant shortages of deposits that meet typical quality standards for mineral processes. One example from the industrial minerals sector is lithium. “Although lithium processing from spodumene has been practiced successfully for some time (Figure 1), in order to meet increasing lithium demand, miners must find a competitive way to process other lithium-bearing ores, such as lepidolite and petalite,” explained Parker.
As a result of this growing need to process more complex ores, “reagents need to be strong, yet selective against penalty elements and gangue materials,” said Guitard. “Lower grades and higher throughputs also necessitate reagents that enable faster value mineral flotation kinetics.”
“Generally, lower ore grades necessitate the processing of more tons to yield the same amount of concentrate,” agreed Parker. “In existing operations, this means that the flotation process must handle more tons within the same amount of time, leading to equipment running beyond design capacity. The flotation process therefore requires reagents that can float faster to deliver the ore to the froth zone quickly, so that it can be swiftly removed. To meet these needs, custom-formulated reagents that improve flotation kinetics, while delivering optimum grade and recovery, are a must.”

laboratory. Photo: Arkema-ArrMaz.
One Ore, Many Products
Miners are also now exploring strategies and technologies to maximize recovery of comingled metals and minerals from the same ore body. These ores “create the greatest challenge for today’s metallurgists,” said Parker.
“In some cases, mica, feldspar and high-purity quartz can be successfully beneficiated from the same deposit with better reagents, equipment and design. High purity tungsten and fluorspar can be produced using custom-designed collectors that achieve higher grade and recovery compared to commodity fatty-acid collectors,” the Arkema-ArrMaz expert continued.
“Understanding the mining process from exploration to production optimization is key to developing reagents that can successfully discover an ore’s true potential, unlocking maximum value throughout the entire beneficiation process.”
A similar trend is seen in metal mines, as Solvay’s Guitard noted, with tailored reagents designed to maximize the extraction of precious metals such as gold, silver and platinum, that can be sold as byproducts. These reagents enable miners to maximize their net smelter returns by capturing maximum value through both precious and base metal recovery.
Safer, More Sustainable Reagents
Enabling the processing of lower-grade, more complex ores is, however, only one side of the coin; on the other side is sustainability. The development of safer reagents that reduce the impact of mining on people and the environment is, therefore, a top priority. For example, there is a movement to replace hazardous chemicals, such as the use of sodium hydrosulfide (NaSH) in copper-molybdenum (Cu-Mo) separation.
“There are several issues with NaSH, including stench, risk of exposure to highly toxic hydrogen sulfide gas, performance deficiencies when treating difficult ores, and logistical challenges in handling large volumes,” explained Solvay’s Guitard. To solve these challenges, Solvay has introduced a depressant technology that can replace up to 60% of the NaSH and is now in commercial use globally. Its latest innovation, now in commercial use at a large copper operation in North America, achieves 100% NaSH replacement at an overall consumption of 1.5 kg/t of molybdenum plant feed, compared with the previous 11.5 kg/t NaSH consumption.
It is often not as easy as simply developing a replacement, however, as Paul D. Slater, vice president of mining – U.S. at SNF, explained: “Alternative chemistry often creates residual effects, creating undesired byproducts, that must also be accounted for, but can be mitigated by working with end-user mining customers to provide a whole system approach solution.”
Water sustainability is another key to sustainable minerals processing.1 Two particular focus areas, according to Parker, are preserving water clarity and protecting water from aquatic toxins.
“Suspended fines often tie off collectors and other surface-active ingredients used in flotation; water free of clay and turbidity, therefore, helps to reduce reagent consumption,” the Arkema-ArrMaz expert noted. Improving water clarity with flocculants is a balancing act, however, as the cost must be weighed against gains in mineral grade and recovery.
“An application system that dynamically handles changes in flotation demands connected to water turbidity can maximize water quality at the lowest cost possible,” said the Arkema-ArrMaz expert. “Wise stewardship and custom flocculant application systems, designed for each mine’s complex water system, can keep water clean for discharge. Systems that isolate bodies of water in ponds to monitor and treat for clarity and water chemistry add another level of protection.”
When it comes to protecting water from toxins, as the impact of mining chemicals on water quality and aquatic life is better understood, reagent suppliers are increasingly able to use libraries and chemical test data to develop products that are safer and more sustainable. As part of the development of new, large mines, Parker also advises specific aquatic testing on local fauna and flora at a regional laboratory.
After safety and water use, a third key pillar of sustainability is lowering the carbon intensity of mining. As an example of the role mining chemicals can play here, sodalite scale inhibitors for the Bayer Process in the alumina industry combine sustainability and profitability, explained Ryan Zheng, global marketing manager – alumina and industrial minerals at Solvay.
“Scale accumulation often leads to costly downtime, exposes workers to the acid-based chemicals needed to treat the scale, and reduces refineries’ energy efficiency,” said Zheng. “Sodalite scale inhibitors therefore contribute to significant cost savings, by maintaining high efficiency in heat exchangers, and deliver energy savings, which limit the amount of CO2 emissions released into the atmosphere.2 Improving heat transfer efficiency and limiting the formation of sodalite scale can also save billions of gallons of freshwater annually in alumina refineries across the globe.”

Digitalization
In common with other areas of mining, mining process chemicals is seeing advances in digital technologies related to the application and management of sustainable treatment programs. Indeed, a future of smart mineral processing plants is already here, utilizing fewer operators and relying on remote or virtual optimization. It’s a future where plants use artificial intelligence (AI) to help predict chemical usage rates within specific applications to maximize recovery rates and lower overall operational costs, as well as to determine the ideal type of chemical for a particular type of ore being processed.
However, even in this digital world – where human interface is being replaced by more advanced machine control – the chemical programs, monitoring of the technology and adjustment of the parameters remain ever-important, according to SNF’s Slater, in order to complement and best harness the wave of innovation.
Nicolas Renaud, digital products director – mining solutions at Solvay, agreed. “We see digitalization of mining chemistry as the next phase in mining’s continuous improvement journey,” he explained.
“The choice of chemistry and process conditions are inextricably linked to feed mineralogy. With increasing size, most mines trade the ability to blend ores, requiring concentrators to be adept at managing rapidly changing feed mineralogy. Chemistry is the only lever that can be changed in real time. This can be done by characterizing the feed in real time and leveraging data science algorithms to recommend chemistry and process conditions. Models suggest several percentage points of metals recovery are to be gained in this manner, along with improved water and energy efficiency.”
Incorporating digital technologies into the reagent formulation process also represents a shift in chemical innovation, according to Renaud. The latest technologies can now be harnessed to deliver live formulation of reagent packages: for example, Solvay offers a 5G-enabled dosing system that enables precise blending and dosing control of building-block chemicals during mineral processing trials.
“There are, however, still many hurdles to successfully digitizing mineral processing,” concluded Arkema-ArrMaz’s Parker. “For example, the sensors and analytical devices used must be robust enough to handle the abrasive and harsh conditions found in most mineral processing plants. In addition, getting a representative sample in and out of an analytical device in a dynamically-flowing system presents a significant challenge. Instrument maintenance and calibration in most industrial mineral processing plants is often troublesome, leading to ineffective utilization of the system. Developing equipment and designs to overcome the tough conditions in the plant will further enable the use of digital and AI technologies to control and monitor mineral processing.”
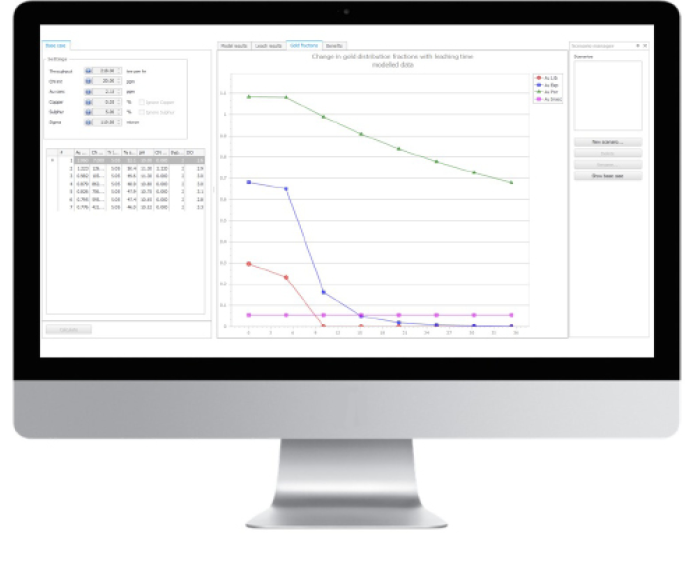
One solution to these challenges is to use historical data, pulled from various sources, to create a model of process conditions that can then be used by metallurgists to see how the process responded to changes – without having to rely on laboratory experiments or plant trials. This approach has been taken by Orica, which offers a software package that can simulate changes to the gold leaching process and present the impact on tails grade (Figure 4). According to the company, by enabling data inputs beyond that of plant data, the overall model fit is significantly improved.
The Way Forward
Mining chemicals suppliers are an innovative bunch – but they don’t work in a vacuum. As Todd Parker of Arkema-ArrMaz concludes, there is a burden on engineering firms, equipment manufacturers and reagent formulators to work together to provide viable solutions. Particularly when developing new, low-grade and complex deposits, miners are “wise to work with reagent formulators, engineering firms and equipment manufacturers to understand the full potential of the ore, and develop a plant design that delivers the highest grade and recovery while keeping costs competitive.”
As one practical example of cross fertilization of innovation, equipment manufacturers are now overcoming the limitations of traditional technology to develop new flotation cells that can recover material previously lost at the very coarse (+300 microns) and fine (-30 microns) ranges during sulfide minerals flotation. In response, Solvay has brought to market a line of reagents to be used with equipment designed to float coarse particles.
“Pilot tests show that these reagents can boost recovery of the coarsest size particles, contributing in a major way to the efficacy of the equipment. New reagents have also been designed to allow flotation of coarser particles in traditional flotation cells (Figure 5),” said Tarun Bhambhani, Principal Scientist, Mineral Processing, Solvay. “In addition, we are studying equipment designed for mitigating losses in the fine size ranges and a product line for these is under development.”

Other areas of innovation include leaching where “improved product development will be required for the harsh conditions associated with extreme pH leaching techniques to provide lower-cost options for the highest-performing separation and dewatering requirements,” said SNF’s Slater. Slater also concludes with an allusion to collaboration between engineering and chemistry – this time in terms of developing dry and water-limited processing techniques: “the continued drive to recover all available free water and recycle within processing conditions, without drawing from outside environmental watersheds, requires a mechanical and chemical balance to these circuits in future.”
It seems that in mining – as in life – finding balance is everything.
References
1. See also: Rowland, J., “Towards a waterless mine”, North American Mining (June 2021), pp. 24-27.
2. According to Solvay, its MAX HT product can deliver savings of 0.1-0.5 tonnes of steam and 0.25-1.25 million BTU per tonne of alumina, eliminated up to 90 kg of CO2 per tonne of alumina. As a result, across the alumina production plants where MAX HT is used, about 2 million tonnes of CO2 are saved, equal to the emissions of about 115,000 American citizens.
